Hypothesis driven research
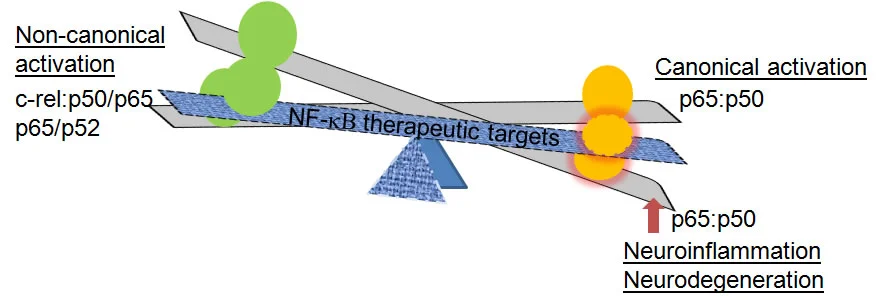
The trajectory of neuroinflammation and neurodegeneration are significantly interlinked in Alzheimer’s disease pathology. The transcription factor, nuclear factor-kB (NF-kB p65) is the master regulator of inflammation. NF-kβ p65 also increases activation of BACE-1, the enzyme critical for amyloid-beta formation. Increased activation of the NF-kB p65:p50 dimers enhance BACE-1 transcription, Aβ genesis, upregulate transactivation of inflammatory and apoptotic mediators that culminate in neurodegeneration. Inhibition of p65 will edge the disrupted balance towards homeostasis, suppress inflammation and neurodegeneration.
The interaction between the inflammatory NF-kB and the anti-inflammatory glucocorticoid signaling pathways is mediated via protein complexes. The NF-kB p65 has a proximal domain that promotes dimer formation, a nuclear localization sequence that allows nuclear translocation and a distal transactivation domain (TAD) that mediates transactivation of inflammatory and apoptosis mediators. Recently, a protein upregulated by glucocorticoids has been shown to mediate many of the beneficial effects of steroids by physically binding and thereby inhibiting the transactivation potential of p65. Using computational simulation scientists, at Provaidya have developed analogs of this NF-κΒ interacting protein, referred to as PGA that sequesters activated p65 and suppresses pathology of neurodegeneration.